Behold the future, here today. Like it did with hybrid cars in 1997, Toyota is at the forefront of a revolution in vehicle technology. The Mirai is Toyota’s first mass-produced fuel cell vehicle, as close to the ultimate eco-car as we’ve got so far, and a vital step in solving the energy demands and emissions issues that come with traditional petrol and diesel-fuelled vehicles.
Yet the new Mirai fuel cell vehicle is much more than the realisation of cutting-edge science theory. But how do fuel cell vehicles work?

What is a fuel cell?
A fuel cell generates electricity by forcing a fuel to react with oxygen. The most commonly-used fuel today is hydrogen, but fuel cells can use almost any hydrocarbon including gas and alcohol. A constant supply of fuel and oxygen is required to sustain the reaction that produces the electricity.
The idea of fuel cells is not a new one – the first examples were designed in the mid-1800s. However, it wasn’t until NASA’s Project Apollo in the 1960s that the idea got off the ground – literally!
What’s fantastic about fuel cells is that the highly efficient process of generating electricity produces no harmful emissions, just lots of pure water making it an environmentally friendly technology. That’s great news if you’re running one inside a spacecraft, but even better here on Earth where fuel cells are a real and viable approach to achieving sustainable mobility. That’s why Toyota is striving to make fuel cell vehicles widely available as soon as possible.
In essence, fuel cell vehicles work by combining the fuel cell’s output with existing and proven hybrid battery technology like those used in the Toyota Prius. Toyota’s world-leading Hybrid Synergy Drive technology underpins its development of fuel cell technology, with the hydrogen fuel cell effectively replacing the internal combustion engine as the main power source. An easy-to-understand explanation of Hybrid Synergy Drive can be found by following this link.
With that understood, let’s explore the functions of the two primary components in a fuel cell vehicle.
Fuel Cell
Electricity is generated in a fuel cell through a chemical reaction between the hydrogen fuel and oxygen. This happens by supplying hydrogen to the anode, or negative, electrode and ambient air to the cathode, or positive electrode.
The fuel cell as a whole is made up of individual cells within a membrane electrode assembly (MEA) that are sandwiched between separators. The MEA consists of a polymer electrolyte membrane with positive and negative catalyst layers on either side. Each cell produces less than one volt of electricity, so hundreds of cells are connected in series to produce the required output voltage. As a whole, the combined body of cells is called a stack, or more commonly, the fuel cell unit.
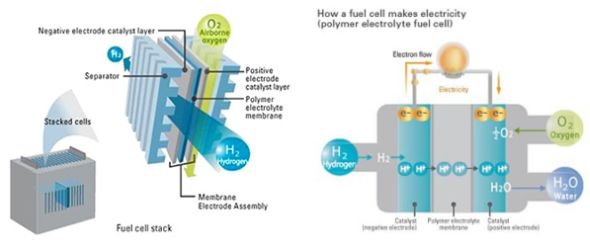
Though a fuel cell can use almost any hydrocarbon as its fuel, hydrogen’s advantage is its high energy efficiency. As it can produce electricity without energy-wasting combustion, it’s possible to convert 83 per cent of the energy in a hydrogen molecule into electricity – that’s more than double the energy efficiency of a petrol engine.
High-pressure hydrogen tank
The hydrogen fuel is stored in two high-pressure (70MPa/700bar) tanks like the one shown below. It uses an innermost layer made of a special polyamide resin layer that is incredibly strong and superbly resistant to hydrogen permeation. This latter quality is especially important as hydrogen molecules are the smallest known to science and are particularly good at escaping through lesser materials!

Toyota has used other materials and techniques to enable it to increase tank capacity and reduce weight – historically, most fuel cell vehicles have needed four separate tanks to provide a useable range. These include the winding angle, tension, volume and wall thickness of the carbon fibre Toyota uses in the outer shell.
How a hydrogen fuel cell generates electricity
The following step-by-step explanation describes how a fuel cell generates the electricity used to power the vehicle:
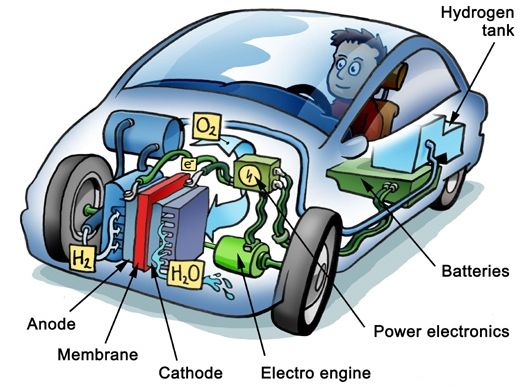
1. Hydrogen is supplied to the anode (negative) side of the fuel cell
2. Hydrogen molecules activated by the anode catalyst release electrons
3. The electrons travel from the anode to the cathode (positive) side of the fuel cell, creating an electrical current
4. The hydrogen molecules that released the electrons become hydrogen ions and move through the polymer electrolyte membrane to the cathode side
5. Hydrogen ions bond with ambient oxygen and electrons on the cathode catalyst to form water
6. Electricity produced by the fuel cell is directed to an electric motor to deliver drive to the vehicle
7. Regenerated energy is stored in a secondary battery that supplements power from the fuel cell by providing auxiliary power to the electric motor
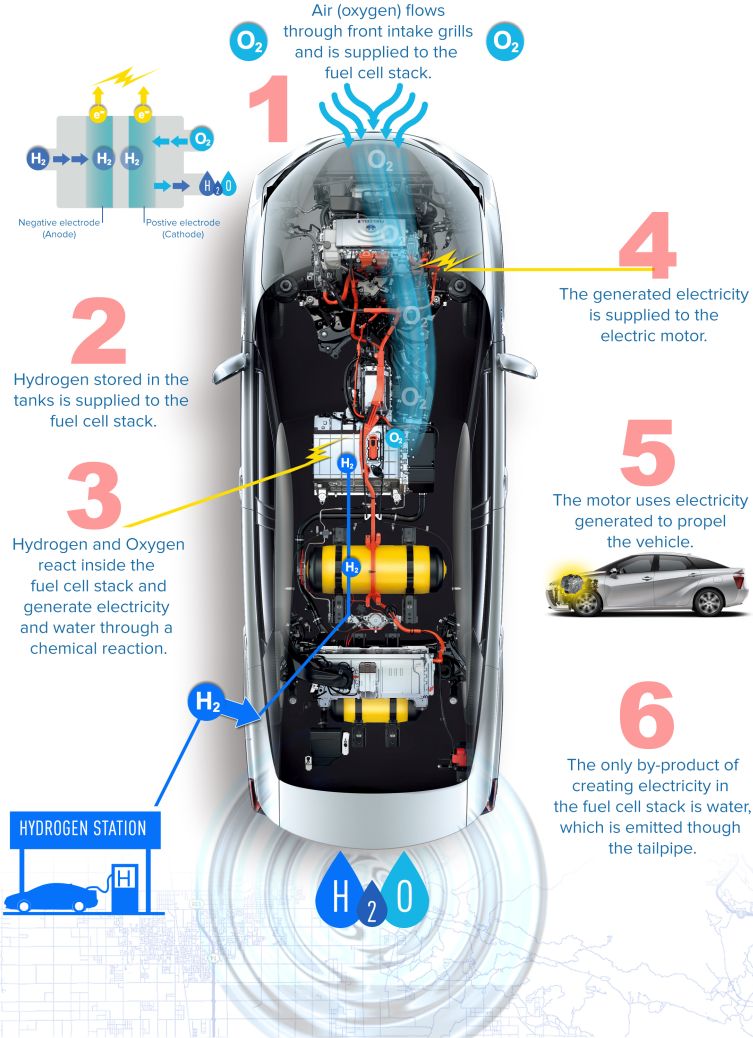
The Toyota FCV Concept
Toyota began researching fuel cell technology in 1992, with its development culminating in the practical FCV Concept that debuted at the 2013 Tokyo Motor Show. Producing no emissions other than water by converting hydrogen into electricity, the FCV Concept promised a range of over 300 miles. It could also produce enough power to provide a Japanese home with over a week’s worth of electricity.

The FCV Concept became a production reality with the launch of the Toyota Mirai in November 2014, and the Mirai is set to go on sale in the UK in September 2015 to selected fleet partners who can utilise the existing hydrogen refuelling infrastructure. Wider availability will depend on the Government’s £11m infrastructure investment program.
More information on the revolutionary Toyota Mirai and the technology behind it can be found at the following links:







Wow! Congratulations it’s taken over a100 years to move away from the combustion engine we have been very very slow on inavation but finally Toyota is leading the world.
This is fantastic news!!!!
I do hope this is something that comes to fruitition as the biggest problem with an electric vehicle is its limited range, this solution would solve that and be environmentally friendly – providing the cost of purchasing such a vehicle is the same as a petrol engine…
This could revolutionise the car completely, and is long overdue
Hello Anthony
Thanks for your post.
We plan to bring a fuel cell vehicle to market in 2015 so it is definitely on the way! The cost will be more expensive than a petrol engine but as we know economies of scale and time can help bring prices down.
Hope this helps
David,
Thanks for letting me know.
Logistically speaking, how will Toyota combat the current issue that there is no network of Hydrogen fuelling stations – is there any plans for major companies such as BP, ESSO, and even supermarket chains to introduce these to their networks? And how will the cost for such “fuel” be calculated, I would expect this to be a fraction of the current petrol prices?
I appreciate pricing would be a sensitive area, and they would not be released until the car is due for launch, but in terms of manufacturing costs etc what percentage higher are these vehicles costs to make vs an equivalent petrol engine?
In the next year or two we are planning on replacing our 2 cars, one of the biggest issues we have with an electric vehicle such as the Nissan Leaf is the restricted range of the car per charge, factor in the monthly cost to “rent” the battery and the cost of the vehicle and I find these cars loose their cost cutting appeal very quickly.
Would be keen to know what might be our next family vehicles, whether it be an electric car with a bigger range or a hybrid vehicle
Hi Anthony
Thanks for your reply, this is a great topic to discuss.
You hit the nail on the head as the infrastructure will undoubtedly be the biggest hurdle to overcome. We know we can make fuel cell cars (we have been doing this for many years) and we know they work. The market though now needs to evolve to support these vehicles which is why we and a number of other companies and manufacturers are members of the H2 Mobility Project to understand the infrastructure involved in this developing new technology. Issues such as the production of hydrogen as well as the transportation all need to be addressed. In terms of the price of hydrogen it is still too soon to say what this is likely to be but it is an exciting time with the development of this technology.
Taking your point about your next vehicle, your comments are exactly why we believe Plug-in hybrid technology is the next step until we reach the ultimate eco-car which is hydrogen. Plug-in provides the benefits of both hybrid and electric vehicles but with none of the downsides associated with full EV’s such as range anxiety. With a Plug-in you are not restricted on charging time or having to wait for this, nor do you need to plan your journey via charging points. Once the electric charge has been used the vehicle reverts to driving as per a normal hybrid.
Let us know if you have nay further questions or would like more information.
Thanks for the info, i will read up on the other blog you mentioned.
It really is a facinating step in car evolution – especially when you think what the future of these vehicles will look like. hybrid seems the logical choice for the moment and Toyota do seem to be at the forefront of this technology which is brilliant!
Could you tell me please as hydrogen is volatile will the tank and engine have to be Chechen frequently, and how often please. Thank you. How much will this cost and when will it be out
Hi Sherrie,
The tank and engine would be checked at evert service.
In terms of cost, what parts are you referring to?
Thanks.
HYDROGEN STORAGE IS THE PART I TRIP OVER. THAT THE HYDROGEN PRESSURE CONTRIBUTES TO A FEAR FACTOR IS DEMONSTRATED BY THE CONVENTION ADOPTED TO GIVE THIS PRESSURE IN ‘ATMOSPHERES’ RATHER THAN PSI-NUMERICALLY 15 TIMES HIGHER. THIS ALLOW THEM TO NOT MENTION NUMBERS GREATER THAN 10,000 PSI. THE ‘POWDERING’ BEHAVIOR SAID TO ABSORB THE ENERGY OF AN EXPLOSION IS A FANTASY MEANT TO DISTRACT THE USER TO THE REALITY OF HYDROGEN GAS STORAGE. THE PUBLIC’S SUSCEPTIBILITY TO ARGUMENTS OF THIS TYPE IS DEMONSTRATED BY ITS WILLINGNESS TO TRAVEL IN A VEHICLES CARRYING GALLONS OF GASOLINE. I LIKE THE METAL HYDRIDE ADSORPTION SYSTEMS BETTER IF A CONSUMER ORIENTED DESIGN CAN BE MARKETED.
Thanks for your feedback Dan.
Safety will of course be a top priority as it is for the cars that we sell today. We are part of the H2 Mobility project and more information regarding hydrogen and safety can be found here. http://www.ukh2mobility.co.uk/faqs/
Hope this helps.
**** Why the Hydrogen Fuel Cell Range Advantage Doesn’t Matter and
**** Why chances are good it will be a still-born child..! ****
The Toyota Mirai hydrogen fuel cell vehicle made its official world debut at the Los Angeles Auto Show. Promising 300 miles of driving range between five-minute fill-ups, the Toyota Mirai seemingly overcomes the biggest hurdles of electric vehicles. But what good is a 300-mile range if there aren’t enough filling-stations?
Right now there are just 15 public hydrogen fuelling stations in the U.S. and a handful in the whole of Europe. While Toyota plans to increase that number via partnerships in California and the Northeast, the fact of the matter is that buyers of the Mirai are going to be much more limited in where they can travel. Any EV owner will tell you that depending on where you live, finding a public charging port can be an adventure.
But in an emergency situation, an electric car can be plugged into any grounded 120-volt of 220-volt socket, something not feasible for hydrogen vehicles. With a growing number of EV owners there are also a growing number of privately-owned Level 2 chargers at many homes and businesses across the nation, and the ever-growing number of Tesla Superchargers means Model S owners can cross the country, for free, while never having to wait more than an hour to charge up. Hydrogen cars are years and years away from being able to cross the country in any amount of time.
On paper, 300+ miles of driving range sounds good, but the reality is that it’s more like 150 miles until more hydrogen stations are up and running. Sure, refuelling in as little as three minutes is nice and all, but the fact remains that hydrogen fuel cell vehicles are going to be even more limited in where they can travel than a lot of plug-in vehicles. By comparison, there are now literally thousands of electric car charging stations spread out across nearly every state in the union. Finding a place to plug-in isn’t nearly as big a deal as it was just four years ago, and with Level 2 charging stations now costing as little as $500, the number of places to plug-in is growing exponentially.
A single hydrogen fuel site on the other hand costs about $1.4 million each, which is the primary reason they’re few and far between. To replace the more than 100,000 gasoline stations with hydrogen fuel would cost hundreds of billions of dollars to serve a fuel that, at best, will be on price parity with gasoline. As it stands, 300 miles worth of hydrogen gas will cost $50, though Toyota is willing to pick up the tab for up to the first three years after purchase. Hyundai on the other hand wants Big Oil to pick up the tab for building hydrogen stations because they know it’s going to cost a fortune.
So they’ve got that going for them, which is nice. But how long will it take until a nationwide network of hydrogen stations is up and running? By Toyota’s own estimates, they hope to have about 3,000 FCVs on the road by the end of 2017, along with 48 new fuelling stations in California and another 12 in the Northeast by the end of 2016. Yet Tesla already has over 100 Superchargers online in the U.S. alone, and spread out in a much more usable fashion rather than clustered around a couple of population centres. Nissan just signed a partnership giving LEAF owners access to thousands of charging stations for free. Hydrogen has some huge hurdles to overcome just to catch up to electric cars, never mind conventional vehicles.
How long does Toyota think it’ll take to bring hydrogen cars to the masses? The Japanese automaker plans to deliver “tens of thousands” of FCVs by the 2020s, but that’s not even a drop in the bucket in a global car market that already exceeds 80 million vehicles. Yet today in every state in the U.S. you can walk into almost any Chevy or Nissan dealerships and buy a plug-in car. The same is also true for Tesla Motors, except in a select few states that hate freedom.
Even once Toyota gets its near-term infrastructure up and running, drivers won’t be able to go much further north than Carson City or too far west of Pam Springs. Toyota just sponsored an advertisement making fun of the BMW i3 for being unable to complete a trip from Van Nuys to Las Vegas; the Mirai make it there, but it sure as hell won’t make it back home, because the one hydrogen station in Sin City isn’t open to the public.
Finally there’s the cost of hydrogen fuel cell vehicles. Even at $59,000, Toyota is losing a ton of money on each hydrogen car it sells…perhaps as much as $100,000 per vehicle. Tesla is grossing as much as 25% profit on each Model S sold though, and in the world of auto manufacturing, financing a money-losing vehicle is only tolerated for so long.
GM reportedly broke even on the Chevy Volt, and Nissan is nearing profitability with the LEAF; but for Toyota, making so much as a dime on hydrogen fuel cell vehicles is a distant possibility at best. Factor in the cost of building a non-existent infrastructure and decade’s worth of research costs, and Toyota basically needs to hit a homerun for hydrogen for it to stand any hope in an increasingly competitive and efficient new car market.
By far though the biggest competitor for hydrogen fuel cell vehicles are conventional gas cars. Hybrid sales are stumbling in the face of more efficient engines, which hurts Toyota in particular, and with gas prices going even lower the disadvantages of hydrogen grow more apparent; even with free fuel; the “payback” date for the Mirai compared to a conventional sedan with so-so gas mileage must measure in the decades.
But unless gas prices drop down to $1 a gallon, electric cars will still have a tremendous advantage in fuel costs over both gasoline and hydrogen cars. That leaves only “true believers” in hydrogen fuel cell vehicles as potential customers, as the price tag is far too high for a middle class family to “experiment” with. But what if those true believers knew that the hydrogen fuel was probably produced from natural gas? Not exactly a zero emissions car anymore, is it?
Until such a time as hydrogen cars can travel beyond the state they were purchased, the total range Toyota and other automakers make doesn’t matter so much. That means hydrogen fuel cell vehicles really only have one advantage, a refill time of between 3 and 5 minutes. At a Level 3 charger cars like the Nissan LEAF and BMW i3 can get an 80% charge in as little as 30 minutes, but that’s still six to ten times longer than hydrogen cars take.
But everyday battery and -charger technology chips away at that advantage and with a little planning most EV owners never have to plug-in anywhere but home.
After decades of false starts and broken promises, hydrogen cars are finally hitting the road, but in a way that reminds me the 1669 EV1 debacle…
Hello Walter
Thanks for your further post about our fuel cell vehicle.
As we have said previously, we are not anti EV and fuel cell simply represents another avenue of technology for us. We also do not deny that the infrastructure needs to be in place and different markets around the world will develop at different speeds. This is a new technology and we have to start somewhere! It was a similar situation when we introduced the Prius Plug in Hybrid, there were limited charging posts but the introduction of Plug in cars and EV’s has helped the increase the availability. The same will happen with the introduction of the fuel cell. The car is here and this technology will not be going away. It is supported by Governments and industry in the UK, Germany and Denmark and fuel cell will help us reduce our reliance on fossil fuels. It is also possible for hydrogen to be produced with zero emissions and research will continue on this subject.
Thank you Toyota. It’s extremely rare when a large corporation has the moral courage to do what’s right for the human race. I’m looking forward to having an electrolyzer in my garage, hooked up to my solar panels to fuel 2 toyota fuel cell vehicles (I currrently have two toyotas in there now). Keep going Toyota, there’s alot of people that want this vehicle, there is a world that needs this vehicle. -CG in Phoenix, AZ
I’ve been wondering for a while how a fuel cell vehicle works. Thank you for letting me know that the fuel cell is made up of individual cells within a MEA. I find your information about how it’s possible to covert 83 percent of the energy in hydrogen molecule into electric. I’ll need to research more about other equipment that uses fuel cells. I love the idea of being more energy efficient!
Hi Lilia,
Thanks for your kind words about this blog post.
We are really glad you found it helpful.
Good luck with your extra research!
Thanks.
I drive from England to Scotland regularly and I spend time in the Highlands on single track roads.
I have been using Toyota Hybrid Cars for six years plus and now have a RAV 4 Hybrid which satisfies much of my requirements. It is good for all weathers, comfortable, powerful and fuel efficient. Having a hybrid in the Highlands means less worry about filling stations and they already have EV chargers in strategic areas but, in an area where, if you break down, recovery vehicles have a long way to come, the dual fuel system gives greater confidence. I am waiting for a hydrogen fuelled SUV that has the capabilities of a RAV 4 both on road and off road. There is no better area to test that than the Highlands of Scotland.
Dreaming of the day I can just do that
Hi Garth,
Thanks for the comment. You’re right, the RAV4 Hybrid is a very capable car! We hope to be able to bring you a hydrogen 4×4 for your journeys.
Thanks